 What is Chloroform?
What is Chloroform?
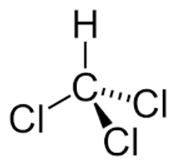
Chloroform is a colorless, volatile, liquid derivative of trichloroethane with an ether-like odor.
Uses
Formerly used as an inhaled anesthetic during surgery, the primary use of chloroform today is in industry, where it is used as a solvent and in the production of the refrigerant freon.
Sources & Potential Exposure
Chloroform may be released to the air as a result of its formation in the chlorination of drinking water, wastewater and swimming pools. Other sources include pulp and paper mills, hazardous waste sites, and sanitary landfills. The major effect from acute (short-term) inhalation exposure to chloroform is central nervous system depression. Chronic (long-term) exposure to chloroform by inhalation in humans has resulted in effects on the liver, including hepatitis and jaundice, and central nervous system effects, such as depression and irritability. Chloroform has been shown to be carcinogenic in animals after oral exposure, resulting in an increase in kidney and liver tumors.Chloroform may be released to the air from many sources related to its manufacture and use, as well as its formation in the chlorination of drinking water, wastewater, and swimming pools. Pulp and paper mills, hazardous waste sites, and sanitary landfills are also sources of air emissions. The background level of chloroform in ambient air in the early 1990s was estimated at 0.00004 parts per million (ppm). Human exposure to chloroform may occur through drinking water, where chloroform is formed as a result of the chlorination of naturally occurring organic materials found in raw water supplies.
Federal Regulations
EPA has classified chloroform as a Group B2, probable human carcinogen.

 Americas
Americas Europe
Europe Français
Français Deutsch
Deutsch Italiano
Italiano Español
Español