 What is Acetone?
What is Acetone?
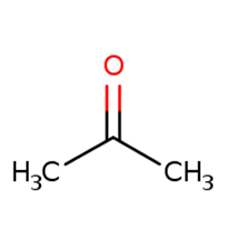
Acetone is a colorless, volatile, flammable organic solvent. Acetone occurs naturally in plants, trees, forest fires, vehicle exhaust and as a breakdown product of animal fat metabolism.
Uses
Acetone is an important commercial chemical that is used as a solvent in consumer products and in industrial processes. Another major use of acetone is as a starting material to make other chemicals.
Sources & Potential Exposure
Workers making or using acetone may breathe in contaminated air and have skin contact. People using consumer products containing acetone, such as paints and glues, nail polishes, nail polish removers, and some pet care products, also may breathe in contaminated air and have skin contact. Breathing in air contaminated with smoke is another way people can be exposed to acetone. Most acetone released during manufacture or use will enter the air. In air, it can be broken down by light or other chemicals. Acetone moves from air into soil and water by rain and snow. It also will move from water or soil into air. Acetone does not bind to soil or build up in aquatic organisms. It will be broken down rapidly by microorganisms in soil and water.
Federal Regulations
The U.S. EPA IRIS program determined that data were inadequate for an assessment of the human carcinogenic potential of acetone.

 Americas
Americas Europe
Europe Français
Français Deutsch
Deutsch Italiano
Italiano Español
Español