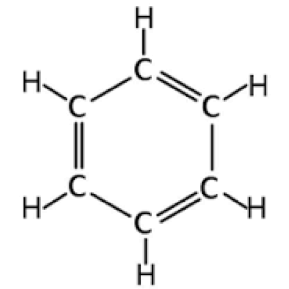
Benzene
 What is Benzene?
What is Benzene?
Benzene is a clear, colorless, highly flammable and volatile, liquid aromatic hydrocarbon with a gasoline-like odor.
Uses
Benzene is used to make plastics, resins, synthetic fibers, rubber lubricants, dyes, detergents, drugs and pesticides. Benzene is also used as a constituent in motor fuels; as a solvent for fats, waxes, resins, oils, inks, paints, plastics, and rubber; in the extraction of oils from seeds and nuts; and in photogravure printing. It is likewise used as a chemical intermediate. Additionally, Benzene can be used in the manufacture of detergents, explosives, pharmaceuticals, and dyestuffs.
Sources and Potential Exposure
Benzene is found in crude oils and as a by-product of oil-refining processes. In industry benzene is used as a solvent, as a chemical intermediate, and is used in the synthesis of numerous chemicals. Benzene is likewise found in emissions from burning coal and oil, motor vehicle exhaust, and evaporation from gasoline service stations and in industrial solvents. These sources contribute to elevated levels of benzene in the ambient air, which may subsequently be breathed by the public. Individuals may be exposed to benzene by consuming contaminated water. Acute (short-term) inhalation exposure of humans to benzene may cause drowsiness, dizziness, headaches, as well as eye, skin, and respiratory tract irritation, and, at high levels, unconsciousness. Chronic (long-term) inhalation exposure has caused various disorders in the blood, including reduced numbers of red blood cells and aplastic anemia, in occupational settings. Reproductive effects have been reported for women exposed by inhalation to high levels, and adverse effects on the developing fetus have been observed in animal tests. Increased incidence of leukemia (cancer of the tissues that form white blood cells) have been observed in humans occupationally exposed to benzene.
Federal Regulations
EPA has classified benzene as known human carcinogen (cancer causing) for all routes of exposure.

 Americas
Americas Europe
Europe Français
Français Deutsch
Deutsch Italiano
Italiano Español
Español